Cancers | Free Full-Text | Real World Outcomes versus Clinical Trial Results of Durvalumab Maintenance in Veterans with Stage III Non-Small Cell Lung Cancer | HTML

Real world data of durvalumab consolidation after chemoradiotherapy in stage III non-small-cell lung cancer - Lung Cancer
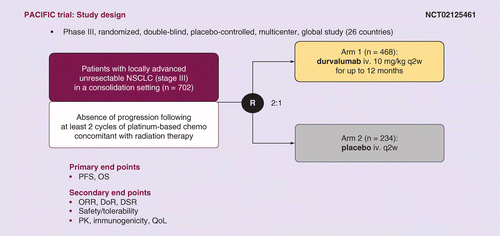
AstraZeneca presents superior progression-free survival for IMFINZI® ( durvalumab) in the PACIFIC trial of patients with locally-advanced unresectable lung cancer at ESMO 2017 Congress

Stage III NSCLC - NSCLC Treatment Paradigm - Text Module - NSCLC Practical Guidance - Oncology - Clinical Care Options

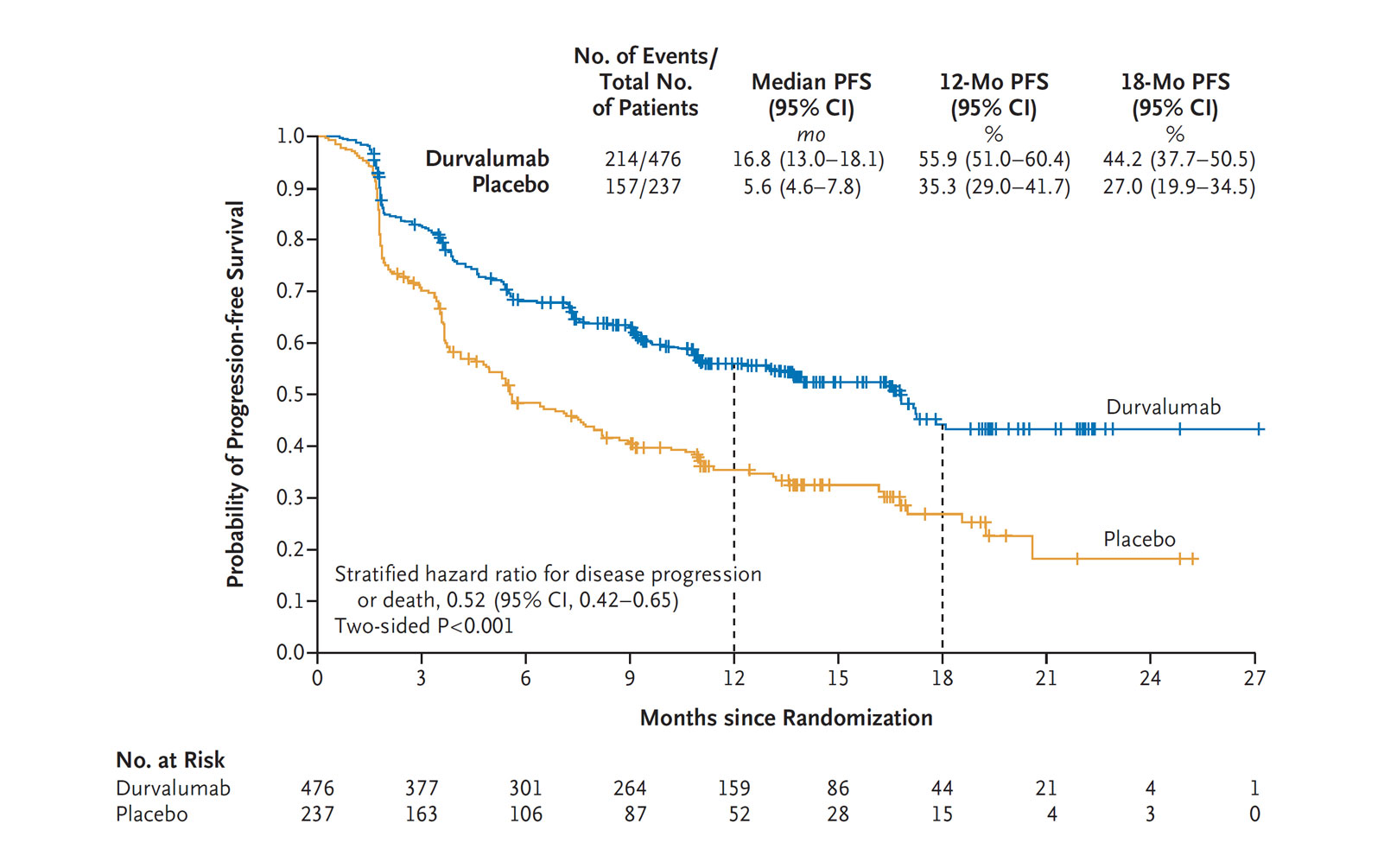
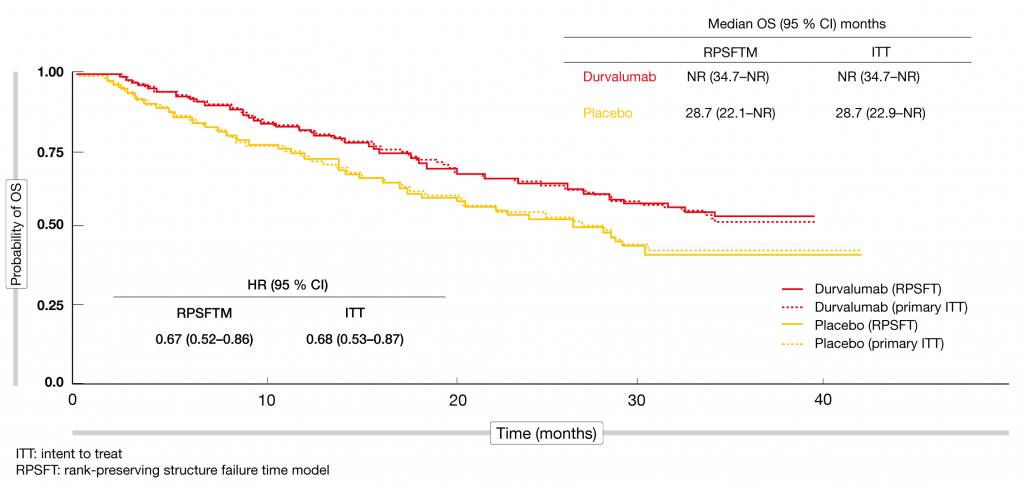
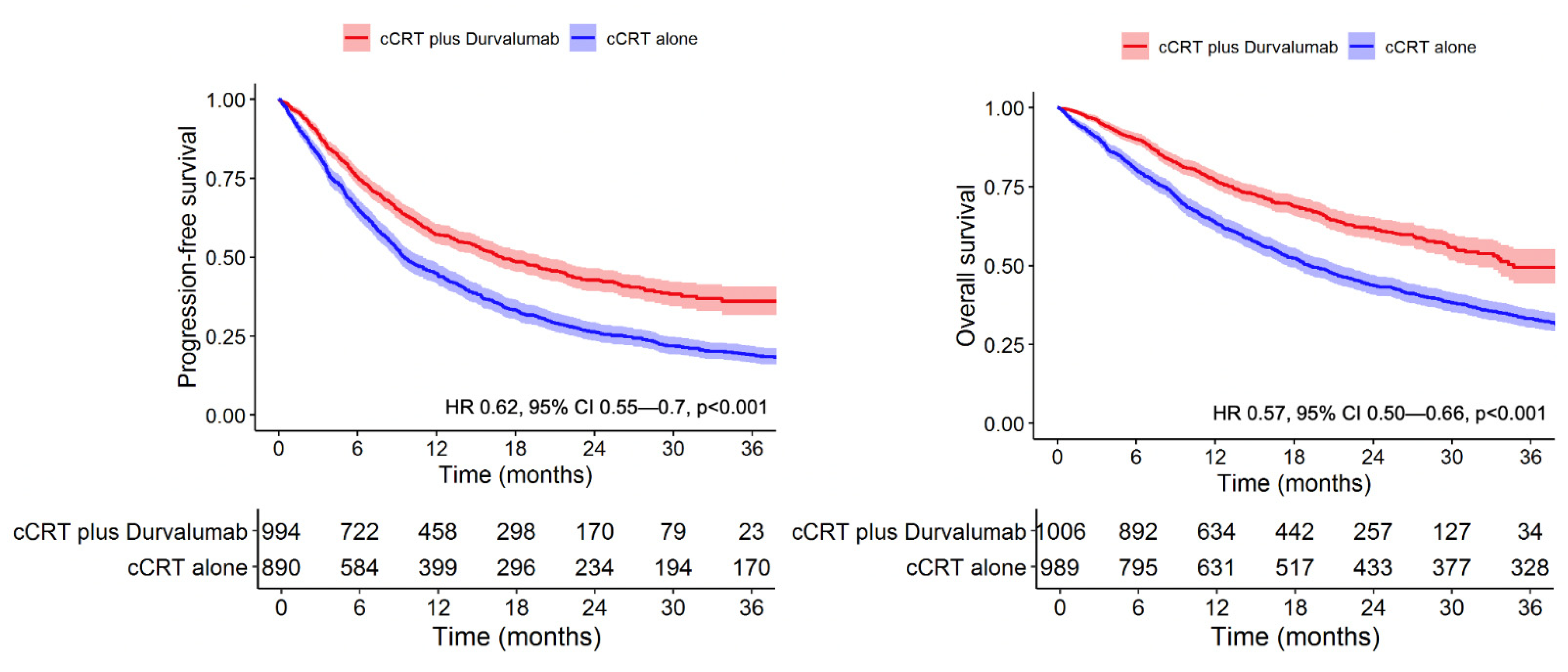
Four-Year Survival With Durvalumab After Chemoradiotherapy in Stage III NSCLC—an Update From the PACIFIC Trial - ScienceDirect

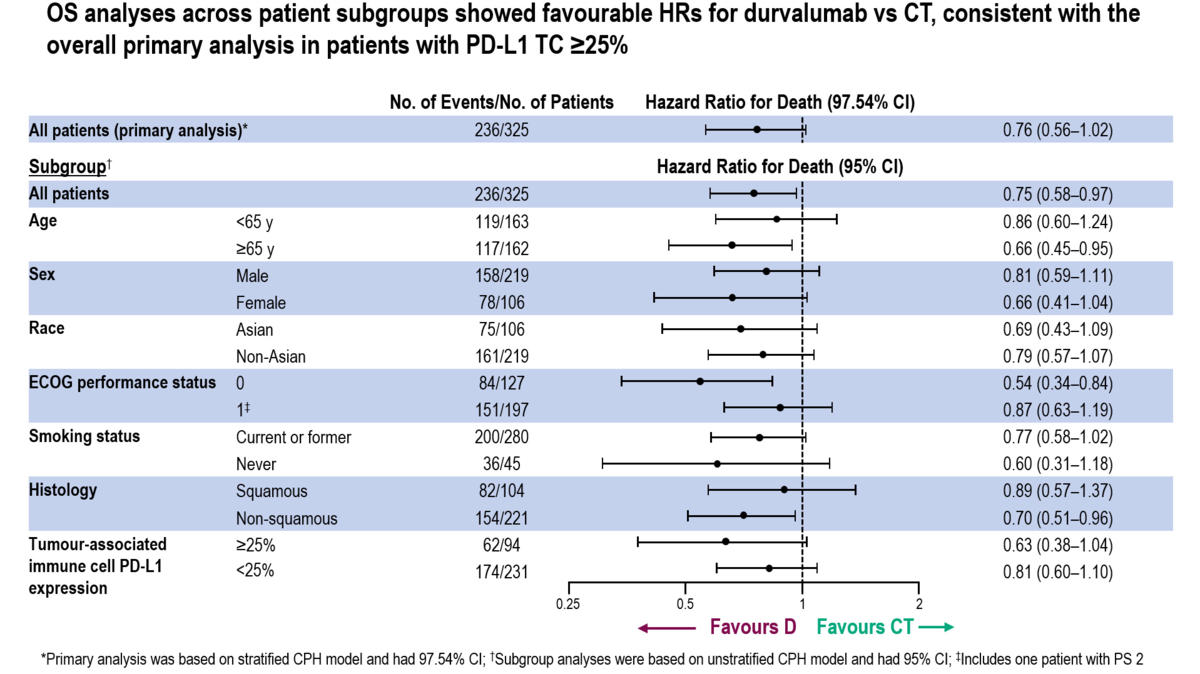
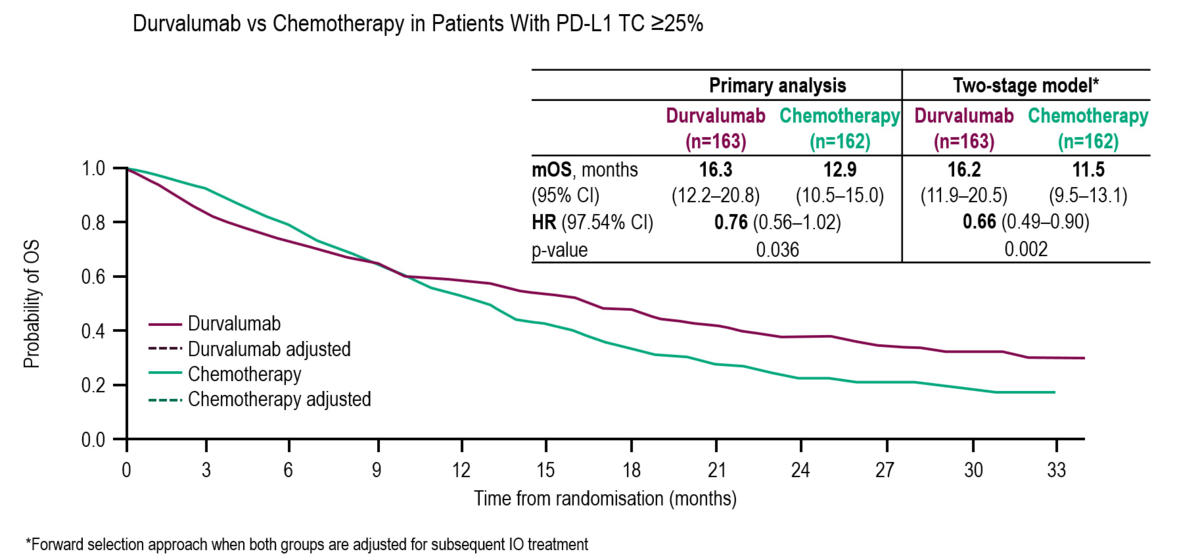
Durvalumab vs placebo consolidation therapy after chemoradiotherapy in stage III non-small-cell lung cancer: An updated PACIFIC trial-based cost-effectiveness analysis - Lung Cancer

PACIFIC 5-Yr Update - Capsule Summary Slidesets - Lung Cancer - 2021 ASCO Virtual Annual Meeting - Oncology - Clinical Care Options

david vicente baz on Twitter: "Very proud of our contribution to PACIFIC clinical trial. Durvalumab as new standard of care in unresectable stage III non small cell lung cancer after chemoradiotherapy Many
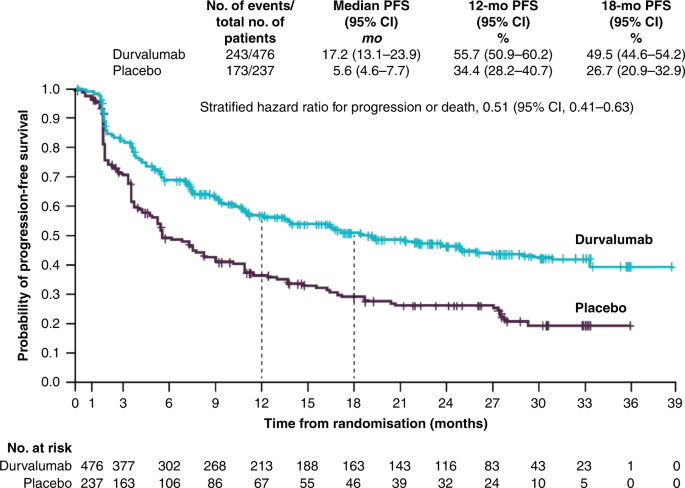
Beyond chemoradiotherapy: improving treatment outcomes for patients with stage III unresectable non-small-cell lung cancer through immuno-oncology and durvalumab (Imfinzi®▽, AstraZeneca UK Limited) | British Journal of Cancer

A Phase III Study of Durvalumab (MEDI4736) With or Without Tremelimumab for Previously Treated Patients With Advanced NSCLC: Rationale and Protocol Design of the ARCTIC Study - Clinical Lung Cancer